“Salt is salt” is a common misconception which has permeated the deicing industry for years. In reality, there are several chemical and physical properties that distinguish different salts from each other. Let’s learn about a specific type of deicer containing complex chlorides and why that matters for road safety, infrastructure, and the environment.

The Chloride Salts
The most common family of deicing ingredients are chloride salts. Chloride salts work by interfering with water molecules’ ability to freeze. They act as freeze point depressants, and each one lowers water’s freezing point to a certain temperature. The chart below reflects the practical or “real world” temperatures of each chloride salt.
- Click here to learn more about the difference between practical temperature and eutectic temperature.
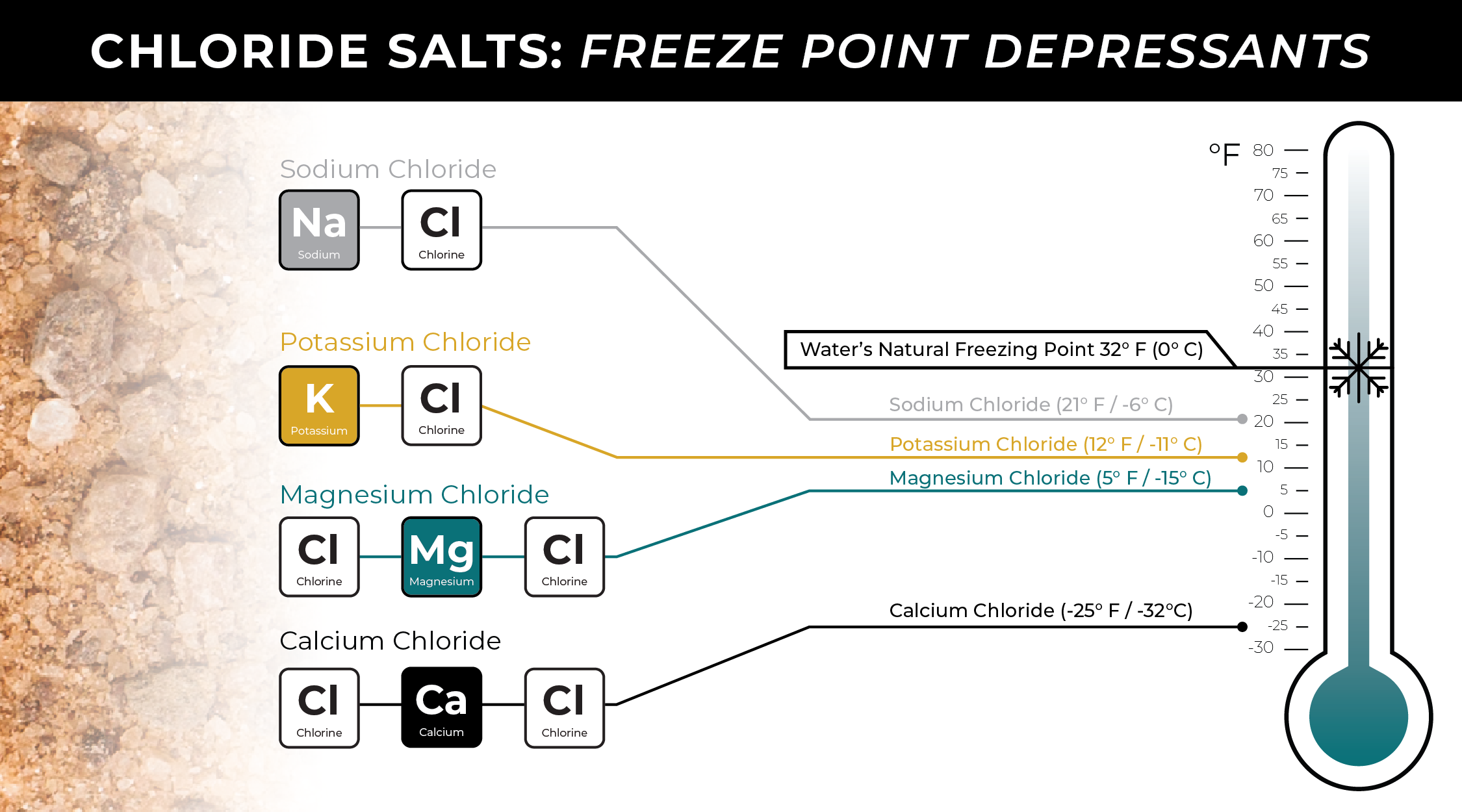
The 4 types of chloride salts used in deicers are:
Sodium Chloride (NaCl)
Sodium chloride, or regular white rock salt, is the most commonly used.
- Sodium chloride lowers water’s freezing point to a practical temperature of about 21° F / -6° C.
Potassium Chloride (KCl)
Potassium chloride is probably the least common chloride to use in deicing.
- Potassium chloride lowers water’s freezing point to a practical temperature of about 12° F / -11° C.
Magnesium Chloride (MgCl2)
Magnesium chloride is less common than sodium chloride and is typically sold as a liquid or added to solid deicers. It has also been found to reduce corrosion.
- Magnesium chloride lowers water’s freezing point to a practical temperature of about -5° F / -15° C.
Calcium Chloride (CaCl2)
Calcium chloride works at the lowest temperatures and is typically used as a liquid additive for some solid deicers.
- Calcium chloride lowers water’s freezing point to a practical temperature of about -25° F / -32° C.
What Are Complex Chlorides?
A complex chloride is a naturally derived, homogenous blend of the 4 main chloride salts. Complex chlorides are mined from ancient sea deposits and also contain other naturally occurring minerals.
Some deicing companies add chlorides together to try and boost performance or reduce corrosion (such as coating sodium chloride granules with liquid magnesium or calcium chloride to work at lower temperatures). These mixtures are NOT complex chlorides. They are not homogenous mixtures, do not have the same chemical and physical properties, or perform the same as a natural blend of complex chlorides with minerals.
Chemical Synergism
Complex chlorides display a natural phenomenon called chemical synergism. That means the combined effect of chemical substances is much greater than the sum of its parts. The substances enhance each other’s efficiency and make their combined efforts even more powerful. Synergism is most commonly noted in the medical community by combining medicines for an even greater effect on the body:
- Example: the combination of aspirin and caffeine for synergistic pain relief

Chemical synergism in a deicer amplifies the performance of each chloride. A blend of 4 different chlorides creates a dynamic solution as each chloride continues to work together for longer lasting performance and prevents re-freezing on road surfaces.
On the other hand, deicers with artificially added chlorides, display the chemical “additive effect.” Their ingredients simply perform their individual chemical reactions without affecting each other’s performance.
Ice Slicer: Complex Chlorides with Minerals in Action
Harvested from a Jurassic Era mineral deposit in central Utah, Ice Slicer is the poster child for powerful complex chlorides. The earth formed this deposit over the span of 200 million years, perfecting its unique blend of chloride salts and over 60 trace minerals; it’s truly mother nature’s perfect recipe for melting ice and snow.
Don’t just take our word for it...let’s hear from “snow boots” on the ground and see what makes the complex chlorides and minerals in Ice Slicer so special.
Complex Chloride Survey
The Western Transportation Institute at Montana State University conducted a thorough study of “the unique synergy of complex chlorides and mineral products.” They surveyed 31 road agencies and winter maintenance experts across 16 states and asked how they felt about using complex chloride mineral deicers, including Ice Slicer.
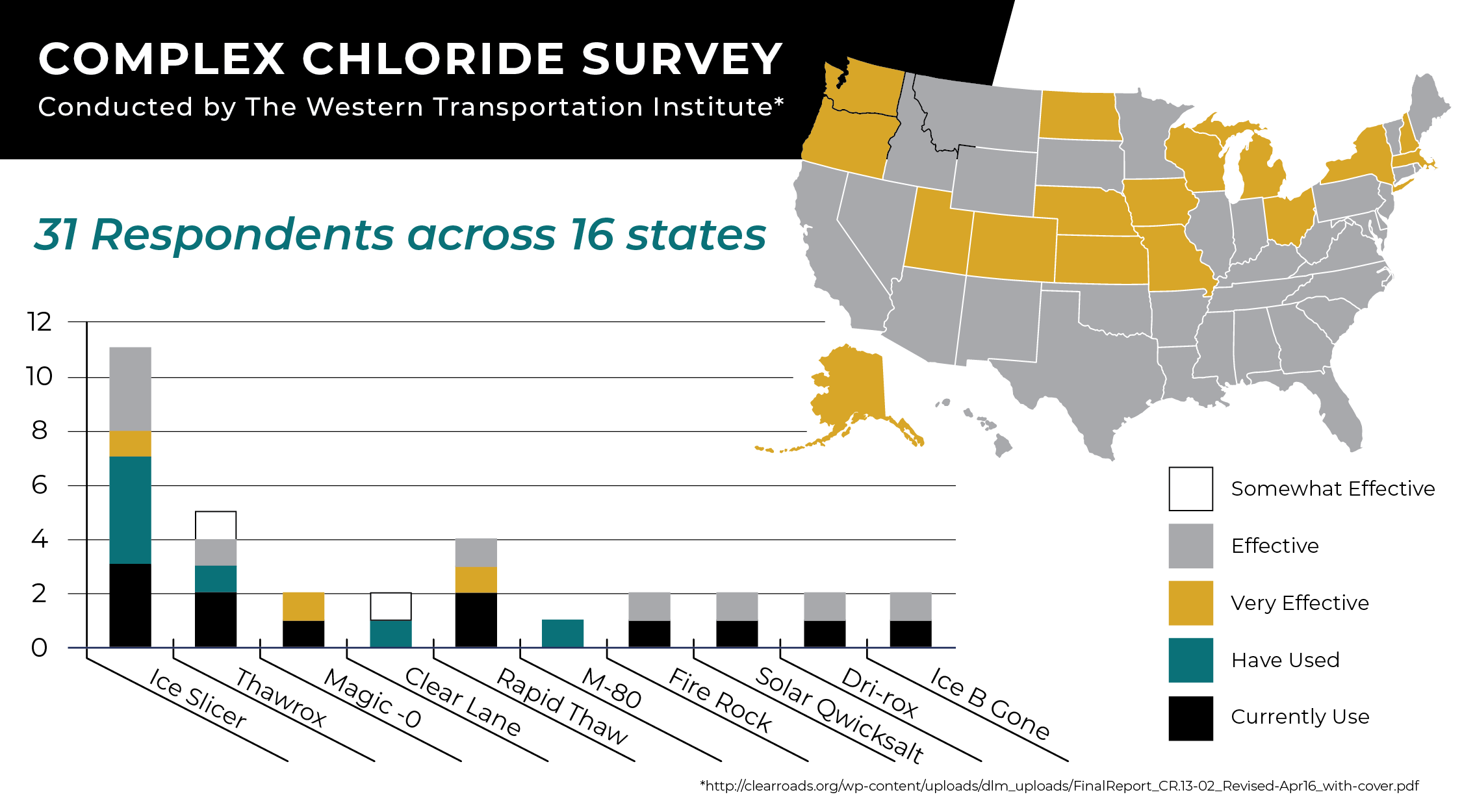
Here are the complex chloride benefits reported by the study’s responders:
- Higher performance:
“The top five primary benefits for using solid complex chloride/mineral products listed by respondents include: 1. “Lowering the freezing point of water” (20.7%), 2. “Prevention of ice formation” (17.2%), 3. “Improving the product longevity on the road” (13.8%), 4. “Preventing refreeze” (13.8%), 5. “Weakening of ice bond to pavement” (13.8%).”
- Reduced infrastructure damage
- Reduced environmental damage
- Financial savings
- Reduction in volume of the material (using less product)
Ice Slicer Testimonials
Ice Slicer users find that complex chlorides and minerals outperform white salt every time. Give us a call before this coming winter to see the Ice Slicer’s unique synergy in action!
“Ice Slicer activates sooner than white salt at any temperature.”
-R. Peterson, Street Super, Nebraska
“We took one truck loaded with white salt and one loaded with Ice Slicer up the freeway...spreading both at the same rate. We turned around at the top - coming back down the Ice Slicer lane was wet - the lane with white salt hadn’t even started melting yet!”
-Heber, Utah
“Roads were usually dry 6-12 hours ahead of other areas when using Ice Slicer!”
-Colorado DOT (Department of Transportation)
“At all temperatures, Ice Slicer was superior to standard road salt at melting snow & ice.”
-Utah Winter Operations
“I have been in the snow and ice control business for over 25 years and am always looking for better and more cost effective ways to deal with our many black ice and freezing rain events. Ice Slicer has outperformed all other brands we have used.”
-Magnum Enterprises, Ketchikan Alaska
“We have found many advantages with Ice Slicer. Most apparent is how much quicker it melts ice compared to white salt. We now have citizens calling to specifically request Ice Slicer in their neighborhoods.”
– Colorado Springs, CO

